1 (888) 801-4801
Bard Rochester - 0115411 - BARD ONFLEX MESH LARGE WITH POCKET 4.0" X 6.2" (10.2CM X 15.7CM)
Prescription or Medical/Professional/ DME License Required. Upload or Send via e-mail.
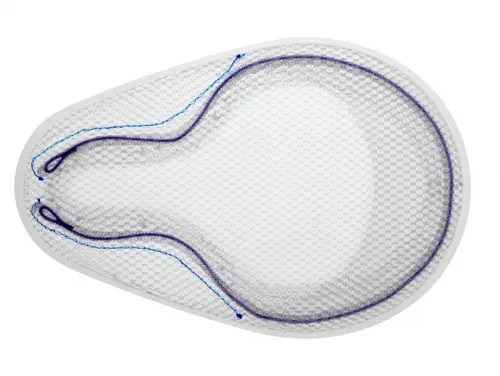
For Open Preperitoneal Inguinal Hernia Repair
Healthcare Professional Downloads:
- Instructions for Use
1 Brown C, Finch J. Which mesh for hernia repair? Annals of The Royal College of Surgeons of England 2010;92(4):272-278.
* Observed in preclinical model, which may not correlate to performance in humans.
Indications
The OnFlex Mesh is indicated for use in the reinforcement of soft tissue where weakness exists, such as in the repair of inguinal hernias.
Contraindications
- Use of this device is contraindicated for infants, children, or pregnant women, whereby future growth will be compromised by use of such mesh material.
- Literature reports that there is a possibility for adhesion formation when polypropylene is placed in direct contact with the bowel or viscera.
Warnings
- The use of any synthetic mesh or patch in a contaminated or infected wound can lead to fistula formation and/or extrusion of the mesh and is not recommended.
- If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. Unresolved infection may require removal of the mesh.
- Do not cut or reshape the Onflex Mesh, except at the opening in the interrupted SorbaFlex PDO monofilament, to accommodate the spermatic cord and outside of the blue limit line in the lateral portion of the mesh, as this could affect its effectiveness. Care should be taken not to cut or nick the SorbaFlex PDO monofilament.
Precautions
Care should be taken not to cut or nick the SorbaFlex PDO monofilament.
Adverse Reactions
Possible complications may include, but are not limited to, seroma, adhesion, hematoma, pain, infection, inflammation, extrusion, erosion, migration, fistula formation and recurrence of the hernia or soft tissue defect. If the SorbaFlex PDO monofilament is cut or damaged, additional complications may include, but are not limited to, bowel or skin perforation and infection.
DAV/OFLX/0917/0027
KEY ADVANTAGES
Lightweight, Large Pore Mesh
- Reduces the amount of foreign material implanted
- Allows good tissue ingrowth1*
- Results in a more flexible and compliant scar formation1
Positioning Pocket
- Medial pocket aids in proper placement
Absorbable SorbaFlex Memory Technology
- Allows the patch to open and conform to the anatomy
- Aids in the proper positioning of the device
Interrupted PDO Monofilament
- Enables easy mesh insertion into the preperitoneal space
Inguinal Notch
- Designed to conform to the iliac vessels
| Manufacturer | Bard Rochester |
|---|---|
| Code | 0115411 |
| Sold By | Each |
| HCPCS | - |
| UPC | - |
| GTIN | - |
| Warehouse | MultiWH |
| WHSXX | GES |

 FSA Eligible
FSA Eligible