1 (888) 801-4801
Quidel - 20196 - QuickVue iFOB Cancer Screening Patient Sample Collection and Screening Kit QuickVue iFOB Colorectal Cancer Screening Fecal Occult Blood Test (iFOB or FIT) Stool Sample 10 Tests CLIA Waived
20196
 FSA Eligible
FSA Eligible

Note:
Please purchase based on product "Code". Images are provided by manufacturers and might not represent the product accurately.
Add to Wish List
Products Details
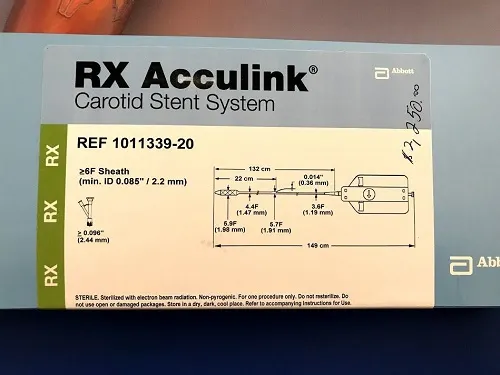
Test Kit, Quickvue Ifob Spcmn Cltn(10ea/kt 12kt/cs
| manufacturer # | 20196 |
| brand | quickvue® Ifob |
| manufacturer | quidel |
| country Of Origin | united States |
Application | Cancer Screening Patient Sample Collection And Screening Kit |
Clia Classification | Clia Waived |
Clia Classified | Clia Waived |
Contents 1 | Specimen Collection Tube, Specimen Collection Paper With Adhesive, Specimen Pouch, Absorbent Sleeve, Return Mailer, Patient Instructions |
Number Of Tests | 10 Tests |
Product Dating | Mckesson Acceptable Dating: We Will Ship >= 180 Days |
Purchase Program Type | Standard Purchase |
Reading Type | Visual Read |
Sample Type | Stool Sample |
Specialty | Immunoassay |
Test Format | Cassette Format |
Test Method | Lateral Flow Method |
Test Name | Fecal Occult Blood Test (ifob Or Fit) |
Test Type | Colorectal Cancer Screening |
Time To Results | 10 Minute Results |
Unspsc Code | 41116120 |
Latex Free Indicator | Not Made With Natural Rubber Latex |
Customer Reviews
Write Your Own Review
Product Information
| Manufacturer | Quidel |
|---|---|
| Code | 20196 |
| HCPCS | - |
| UPC | - |
| GTIN | - |
| Warehouse | MultiWH |
| WHSXX | MKC |
Special Deals